 UCLA and Egyptian scientist accidentally find a new way to bottle stored energy. This missing link for solar energy, hydro and electric cars could be a fast, tiny, biodegradable battery
UCLA and Egyptian scientist accidentally find a new way to bottle stored energy. This missing link for solar energy, hydro and electric cars could be a fast, tiny, biodegradable battery
Penicillin, Teflon, microwave ovens and superglue were all discovered by accident. And now graphene super-capacitors might be the most important accidental discovery of our time – one that can change the way energy is stored. A team of UCLA researchers led by chemist Richard Kaner used a commercial DVD burner to produce sheets of a carbon-based material known as graphene.
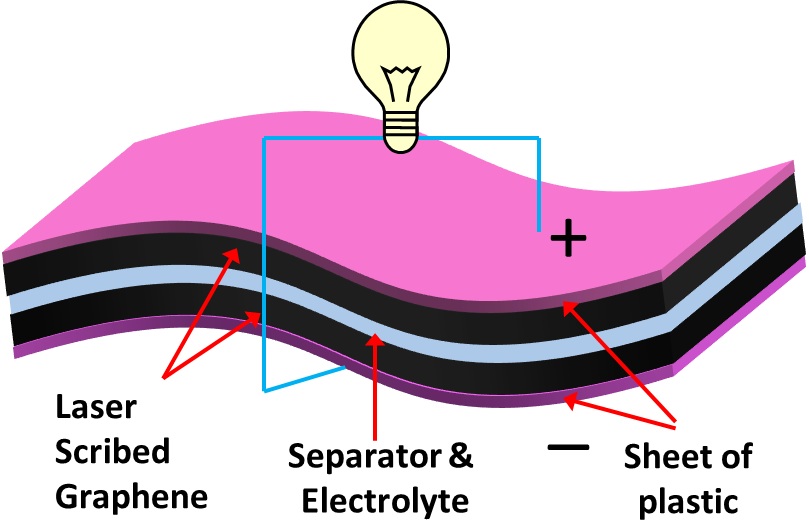
The “accident” occured when Cairo University graduate Maher El-Kady (pictured below) wired a small piece of graphene to an LED and found that it behaved as a super-capacitor, able to store a considerable amount of electricity. Their laser-scribed graphene is ideal as a super capacitor partially because of its enormous surface area, 1520 square meters per gram. Here’s how it works:
The story begins with quirky old kite-flying American, a key and a bolt of lightning. It ends with a jar full of electricity. Benjamin Franklin’s jar of electricity is known as a Leyden jar.
It is a primitive electronic circuit element known as a capacitor. The Leyden jar illustrates some promising characteristics of capacitors. As electrical storage devices, they are extraordinarily simple. You can make one at home with a glass jar and a some aluminum foil.
Capacitors have some advantages over Lithium, Nickle-Metal hydride and other chemical batteries. Batteries convert electrical energy to and from chemical energy. But capacitors store electrical charge by bottling excess electrons on one side of a thin barrier.
So capacitors needn’t contain caustic mixtures of acids, alkalis and toxic metals as batteries do. Capacitors can also be charged many times and they can be charged very fast. Some of the tantalum and electrolytic capacitors inside your computer or iPad are charging and discharging millions of times while you read this.
 If capacitors are so wonderful, why aren’t they used in place of batteries electric cars to laptops and mobile phones?
If capacitors are so wonderful, why aren’t they used in place of batteries electric cars to laptops and mobile phones?
The problem is that capacitors aren’t able to store very much energy. A Lithium Ion battery the size of a Leyden jar can store more than 500,000 times more energy.
But capacitors have improved since the Leyden jar. The graphene capacitor these UCLA scientists created has 4 billion times the capacitence of a Leyden jar.
Since its operating voltage is much lower, it might only store about 40,000 times the energy density of a Leyden jar, but this brings it much closer to the energy density of a chemical battery.
And that could change everything.
A film explaining the story of this invention is a finalist in the General Electric focus forward filmmaker competition.
Watch the film and decide for yourself whether a small, efficient, biodegradable energy storage device might revolutionize the future of energy storage.
Image of graphene supercapacitor from the UCLA newsroom


No worry about big oil. These are storage devices. You must add energy to them to get the energy out. Petroleum based products make use of the energy added to them by natural processes. Oil comes out of the ground with the energy already in it. That’s why it is so hard to replace it.
This concept may one day change the entire energy concept. It’s like putting Nikola Tesla’s theories of “free energy” into actual concrete reality. And we must also thank Ben Franklin for his contribution too.
Incredible. Perhaps these guys should hire security guards to protect their lives as they’ve now developed a huge threat to big oil.
Exciting!